AI And Digital Tools Advance Clinical Trial Supply Chains
By Hiep X. Nguyen, DPharm, Ph.D., DTM

Clinical trials remain the definitive mechanism for validating new therapeutics, yet the supply chains sustaining these trials rank among the most operationally demanding logistics environments in healthcare. The clinical trial supply chain must manage highly unpredictable variables — enrollment rate volatility, protocol amendments, patient dropout, and dose titration changes — while maintaining simultaneous compliance with manufacturing, clinical, and distribution regulations across dozens of countries (Fig. 1).1 The global clinical trial supplies market, valued at approximately $2.73 billion in 2024, is projected to grow at a CAGR of 7.19% through 2030.2 A single day of dosing delay in a Phase 2 or Phase 3 trial costs approximately $40,000 in direct expenditures, with indirect costs from deferred revenue estimated at $800,000 per day.3 The industry median investigational medicinal product (IMP) waste rate remains at 50%, and for sponsors spending approximately $10 billion annually on R&D, optimizing waste could unlock over $100 million in savings.3 On average, bringing a new drug to market requires 10 to 15 years and over $1.5 billion, with approximately 86% of trials failing to meet enrollment timelines.4 The COVID-19 pandemic catalyzed serious investment in AI-driven pharmaceutical supply chain (PSC) resilience, as the sudden demand fluctuations and logistical bottlenecks exposed the structural inadequacy of traditional rule-based planning systems.1 AI-driven predictive analytics now advance demand forecasting, risk management, and operational resilience,5 with platforms such as SAP’s Intelligent Clinical Supply Management solution integrating AI and real-time analytics to optimize supply chains.6 Yet a systematic review of 32 peer-reviewed studies found that the majority of AI/ML applications remain in the theoretical or simulation phase, over 59.3% failed to address regulatory frameworks, and none examined ethical concerns.1
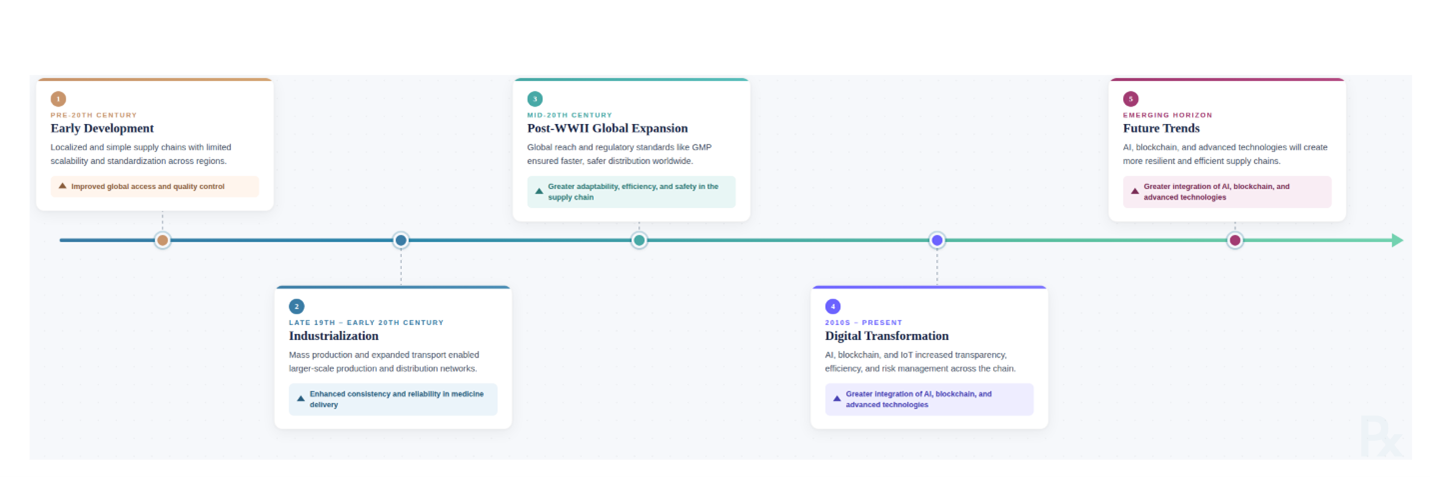
Figure 1. The evolution of pharmaceutical supply chain management.
AI-Driven Demand Forecasting And Inventory Optimization
Demand forecasting in clinical trials is inherently stochastic, driven by enrollment projections, dropout assumptions, and protocol amendment frequency.3 Traditional deterministic tools — exponential smoothing, regression, and time-series analysis — are inadequate for scenarios characterized by significant demand fluctuations.7,8 AI/ML-powered models address these limitations by analyzing historical trends, market shifts, and external factors.1 Among evaluated structures, Long Term-Short Term Memory (LSTM) networks have demonstrated particular promise in fluctuating environments.1 An empirical study of 1.2 million shipments across 39 countries found that Autoregressive Integrated Moving Average (ARIMA) (order 1,0,1) achieved the lowest RMSE (0.0758) but yielded a negative R² (−0.3573), while LSTM (RMSE 0.1589) more faithfully captured nonlinear demand surges.9 Accurate forecasting prevents stockouts — avoiding $50,000 to $100,000 per incident — and reduces excess holding costs by $200,000 to $500,000 annually.9 Risk-based Monte Carlo optimization tools have demonstrated 20%–50% cost savings and 20%–60% drug waste reductions.3 AI-powered supply management software has achieved up to 30% packaging waste reduction and 99% temperature compliance.10 Reinforcement learning (RL) has been applied to inventory replenishment across multiple distribution centers, though inventory optimization was the least explored area, with only two of 32 studies focusing primarily on this topic.1 Real-world deployments include a hospital AI + VMI system that increased efficiency by 42.4% and reduced inventory errors from 0.425% to 0.025%11,12 and Roche’s ML system monitoring sensors across warehousing and production operations.13
Practical Takeaway: Launch a focused pilot within a single study or geographic region by overlaying an AI forecasting model (e.g., LSTM or ARIMA) onto current demand projections. Track forecast precision, stockout frequency, and waste metrics across one to two supply cycles, then use verified performance gains to justify scaling toward global planning integration.
Risk Management, Cold Chain Logistics, And Shipment Mode Optimization
Risk management dominates AI/ML research in PSCs, with 24 of 32 studies focused on risk identification and mitigation.1 AI/ML models enable early detection of vulnerabilities through predictive risk modeling, safety stock management, and backup supplier strategies.1 By 2025–2026, AI platforms will have evolved toward smart control towers that detect interferences and prescribe corrective measures with minimal human intervention.14 Cold chain management is particularly acute given the accelerating shift toward biologics and cell therapies, with cold chain medicines growing at 13% annually between 2017 and 2022.3 Pfizer and Merck have implemented AI solutions for vaccine distribution,15 and Internet of Things (IoT)-enabled sensors integrated with AI analytics generate real-time temperature excursion alerts meeting GMP and GDP guidelines.16 Shipment mode analysis across 1.2 million shipments revealed a significant association between mode and late delivery (χ² = 139.96, p = 3.86 × 10⁻³⁰).9 A PCA–K-Means vendor segmentation methodology identified three performance clusters, with optimized vendor selection projected to reduce logistics costs by 15%–25%.9 Neural networks achieved the best maintenance prediction performance (AUC 96.50%), though class imbalance (90:10 ratio) creates models with high accuracy but lower recall.9
Practical Takeaway: Assemble a streamlined “control tower lite” that consolidates shipment tracking, temperature monitoring, and vendor performance scoring within a unified dashboard featuring automated alerts for delivery delays or thermal excursions. Leverage these real-time signals to reassign shipping lanes and redirect vendor assignments during active study conduct.
Digital Twins, Blockchain, And IoT as Enabling Infrastructure
Digital twin (DT) technology serves as a versatile simulation tool spanning patient-level clinical modeling and supply chain intelligence. In clinical trials, a DT simulates a patient’s physiological characteristics, disease features, and treatment responses through integration of real-world data streams.17,18 A 2025 review proposed an integrated DT hub including clinical care, manufacturing, and logistics domains.19 DTs simulate logistics networks to identify optimal routes and inventory levels, integrating IoT data to monitor temperature and humidity in real time.20 Process analytical technology (PAT)-integrated manufacturing DTs have improved API consistency to 99.95%.21 However, gaps in real-time data and limited generalizability when DTs trained on narrow cohorts are applied broadly remain significant limitations.22,23 Blockchain technology creates an immutable ledger tracking IMP movement from manufacturing through patient administration, with the DSCSA establishing a regulatory precedent for pharmaceutical traceability that informs emerging frameworks for clinical trial supply chains.24 A blockchain-enabled QbD design using Hyperledger Fabric ensures data integrity and dynamic risk control.25 Despite these advances, neither the FDA nor the EMA has developed standardized blockchain adoption frameworks for clinical research.26
Practical Takeaway: Before investing in full-scale digital twin or blockchain solutions, prioritize IoT sensor deployment across high-risk lanes. Use the resulting real-time telemetry to conduct targeted scenario analyses (e.g., alternative routing or depot reallocation) and generate quantifiable evidence that justifies broader technology investment.
Adaptive Trials, AI Agents, LLMs, And Supply Chain Responsiveness
Adaptive trial designs allow real-time protocol modifications, including dosage adjustments and treatment arm changes,27 with reinforcement learning, decision trees, and neural networks continuously updating protocols based on interim results (Table 1).28,29 The stochastic variability in adaptive trials cannot be accommodated by deterministic supply planning.30 AI agents further extend automation: ClinicalAgent improved trial outcome prediction by 0.33 AUC over baseline methods,31 and MAKAR achieved 100% accuracy in patient-trial matching.32 However, misaligned agents that incorrectly forecast arm closures could cause dosing interruptions, making human-in-the-loop oversight a patient safety necessity.33 Natural language programming (NLP) and large language models (LLMs) offer supply chain applications, including anomaly detection in databases and trend identification in inventory systems.34 The C3PO NLP model demonstrated 93% agreement with clinical events committees.35 Application-specific language models (ASLMs), enhanced with retrieval-augmented generation (RAG), outperformed human experts in patient screening (93.6% vs. 85.9% accuracy),36 with RAG-augmented models showing a pooled odds ratio of 1.35 (p = 0.001) over baseline LLMs.37 The connection between protocol quality and supply chain efficiency is direct: well-designed protocols generate predictable enrollment trajectories enabling accurate IMP demand forecasting.38
Practical Takeaway: Synchronize supply planning with clinical operations through a structured review cadence (e.g., biweekly) that converts interim trial data and anticipated protocol modifications into revised supply forecasts. Ensure that all AI-generated outputs undergo cross-functional validation involving clinical, regulatory, and supply chain stakeholders before operational decisions are finalized.
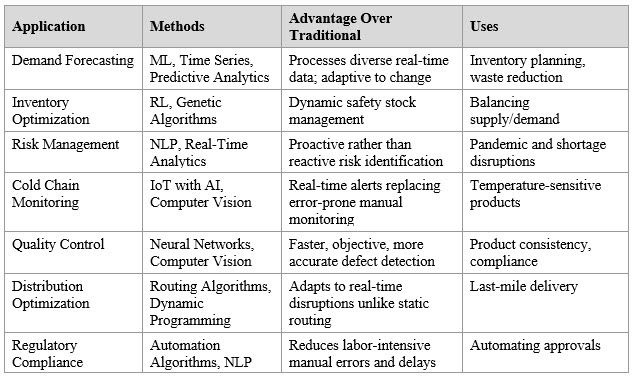
Table 1. Key AI Applications in Pharmaceutical Supply Chain Management
Decentralized Trials And Patient-Centric Supply Chain Models
Decentralized clinical trials (DCTs) require direct-to-patient (DTP) shipment, adding cold chain, compliance, and last-mile complexity.39 By 2025–2026, up to 90% of trials include decentralized components,40 compared with 24% managing DTP shipping in 2016. DCTs offer shorter cycle times, fewer screen failures, and fewer protocol amendments.40,41 Each of the approximately 30% of patient dropouts represents an IMP waste event, and 40% of patients become nonadherent after 150 days.4 Reinforcement learning can leverage continuous data to anticipate and prevent dropout.42 ML-based phenomapping enables adaptive enrichment, reducing sample size by up to 18% while preserving efficacy signals with proportional decreases in IMP procurement and distribution requirements.40,43
Practical Takeaway: For direct-to-patient distribution, initiate a bounded pilot with a defined patient cohort in a single region, collaborating with a qualified logistics partner to validate last-mile delivery reliability, cold chain integrity, and medication adherence monitoring. Confirm operational readiness across these critical parameters before pursuing broader geographic expansion.
Regulatory Compliance, Data Governance, And Ethical Considerations
Among 32 systematically reviewed studies, 19 did not address regulatory concerns, and none examined ethical dimensions of AI/ML in PSC resilience.1 The FDA’s January 2025 draft guidance introduced a risk-based credibility assessment framework for AI models, and the EU AI Act classifies medical AI systems as high-risk, requiring conformity assessment and human oversight.44 Algorithmic bias in supplier selection and opaque automated inventory systems represent concrete clinical trial harms.1 Federated learning preserves patient privacy during collaborative model training,45 while Federated Knowledge Recycling (FedKR) enables synthetic data exchange to minimize re-identification risks.46 Regulatory considerations must be integrated into AI/ML adoption from inception.1
Practical Takeaway: Construct a fit-for-purpose AI governance framework at the outset of each deployment, specifying model objectives, data sources, validation protocols, and designated human oversight responsibilities. Early documentation of these elements ensures audit readiness and positions organizations to meet evolving FDA and EU AI Act requirements proactively.
Strategic Priorities And Future Directions
While numerous AI/ML frameworks have been proposed, most remain in the theoretical or simulation phase.1 Four strategic directions emerge: bridging the implementation gap through field-tested applications and real-time deployment; expanding the methodological toolkit to include reinforcement learning and NLP; incorporating regulatory and ethical dimensions into model design from inception; and extending AI/ML to underexplored areas, including last-mile delivery and patient-centric service.1 The global AI-in-pharmaceutical market is forecasted to reach $16.49 billion by 2034 at a CAGR of 27%,5 and AI is expected to be integrated into 60%–70% of clinical trials by 2030, generating estimated annual savings of $20 billion to $30 billion.47
Practical Takeaway: Anchor the AI implementation roadmap around one or two high-impact use cases (e.g., demand forecasting accuracy or cold chain compliance rates) defined by measurable KPIs. Resist premature portfolio expansion; validate return on investment through documented performance gains before extending AI capabilities to additional supply chain functions. .
Conclusion
The clinical trial supply chain stands at an inflection point. Empirical evidence demonstrates that AI/ML tools can reduce IMP waste by 20%–60%,48 substantially curtail packaging waste, maintain excellent cold chain integrity,10 and significantly lower distribution expenditures through data-driven vendor segmentation.9 Yet the persistent theory-to-practice gap and the absence of ethical and regulatory analysis in published research constrain adoption. Closing this gap requires validated real-world deployment through phased pilots, methodological diversification, regulatory co-development through sandbox collaborations with the FDA and EMA, and equity-centered design serving diverse patient populations. AI and digital tools will redefine the operational standard against which clinical trial supply chain performance is measured — making the difference between trials that deliver lifesaving therapies on time and those that fail patients through preventable logistical shortcomings.
References:
- Al-Hourani, S.; Weraikat, D. A Systematic Review of Artificial Intelligence (AI) and Machine Learning (ML) in Pharmaceutical Supply Chain (PSC) Resilience: Current Trends and Future Directions. Sustainability 2025, 17, doi:10.3390/su17146591.
- Srivastava, P. Clinical Trial Supply in Pharmaceutical Manufacturing: A Comprehensive Guide. PharmaSource 2025.
- Clinical Trial Supply Chain Management: Complete Guide & 2025 Software Directory Available online: https://feeds.vuroyal.com/blog/supply-chain-intelligence-tool-clinical-trial-supplies (accessed on 22 March 2026).
- Harrer, S.; Shah, P.; Antony, B.; Hu, J. Artificial Intelligence for Clinical Trial Design. Trends Pharmacol. Sci. 2019, 40, 577–591, doi:10.1016/j.tips.2019.05.005.
- Vadaga, A.K.; Dokuburra, U.R.; Nekkanti, H.; Gudla, S.S.; Kumari, R.K. Digital Transformation in Pharmaceuticals: The Impact of AI on Supply Chain Management. Intell. Hosp. 2025, 100008.
- lawrence Artificial Intelligence Is Making Clinical Supply Chains More Efficient and More Sustainable: Here’s How. Pharm. Technol. 2025.
- Sah, B.P.; Shirin, B.; Minhazur Rahman, B.; Shahjalal, M. The Role of AI in Promoting Sustainability within the Manufacturing Supply Chain Achieving Lean and Green Objectives. Acad. J. Bus. Adm. Innov. Sustain. 2024, 4, 79–93.
- Saha, R.; Shofiullah, S.; Faysal, S.; Happy, A. Systematic Literature Review on Artificial Intelligence Applications in Supply Chain Demand Forecasting. Available SSRN 5062817 2024.
- Park, K.M.; Pattnaik, S.; Liew, N.; Kundu, T.; Kures, A.O.; Pinsky, E. Smarter Chains, Safer Medicines: From Predictive Failures to Algorithmic Fixes in Global Pharmaceutical Logistics. Forecasting 2025, 7, doi:10.3390/forecast7040078.
- Rai, A. Clinical Trial Supply Management: A Complete Guide Available online: https://clinion.com/insight/what-is-clinical-trial-supply-management/ (accessed on 22 March 2026).
- Mohamadi, N.; Niaki, S.T.A.; Taher, M.; Shavandi, A. An Application of Deep Reinforcement Learning and Vendor-Managed Inventory in Perishable Supply Chain Management. Eng. Appl. Artif. Intell. 2024, 127, 107403.
- Salas-Navarro, K.; Florez, W.F.; Cárdenas-Barrón, L.E. A Vendor-Managed Inventory Model for a Three-Layer Supply Chain Considering Exponential Demand, Imperfect System, and Remanufacturing. Ann. Oper. Res. 2024, 332, 329–371, doi:10.1007/s10479-023-05793-6.
- Kulkov, I. The Role of Artificial Intelligence in Business Transformation: A Case of Pharmaceutical Companies. Technol. Soc. 2021, 66, 101629.
- AI Pharma Supply Chain 2026: Logistics, Inventory Optimization Available online: https://www.pharmafocusamerica.com/articles/ai-pharma-supply-chain-2026-trends (accessed on 22 March 2026).
- Guo, X.; Xie, W.; Li, X. Spatial-Temporal Correlation Neural Network for Long Short-Term Demand Forecasting During COVID-19. IEEE Access 2023, 11, 75573–75586, doi:10.1109/ACCESS.2023.3297143.
- Technology Shaping 2025 Clinical Trial Supply Chain Available online: https://www.precision-globe.com/post/role-of-technology-in-shaping-the-2025-clinical-trial-supply-chain (accessed on 22 March 2026).
- Bruynseels, K.; Santoni de Sio, F.; van den Hoven, J. Digital Twins in Health Care: Ethical Implications of an Emerging Engineering Paradigm. Front. Genet. 2018, 9, 31, doi:10.3389/fgene.2018.00031.
- Sel, K.; Hawkins-Daarud, A.; Chaudhuri, A.; Osman, D.; Bahai, A.; Paydarfar, D.; Willcox, K.; Chung, C.; Jafari, R. Survey and Perspective on Verification, Validation, and Uncertainty Quantification of Digital Twins for Precision Medicine. NPJ Digit. Med. 2025, 8, 40, doi:10.1038/s41746-025-01447-y.
- Wang, Y.; Carter, B.Z.; Li, Z.; Huang, X. Application of Machine Learning Methods in Clinical Trials for Precision Medicine. JAMIA Open 2022, 5, ooab107, doi:10.1093/jamiaopen/ooab107.
- Guibelondo, D.M.T. Digital Twins in Clinical Trials: Revolutionizing Simulation and Logistics Optimization. PharmaFeatures 2025.
- Maharjan, R.; Kim, N.A.; Kim, K.H.; Jeong, S.H. Transformative Roles of Digital Twins from Drug Discovery to Continuous Manufacturing: Pharmaceutical and Biopharmaceutical Perspectives. Int. J. Pharm. X 2025, 10, 100409, doi:10.1016/j.ijpx.2025.100409.
- Badani, A.; de Moraes, F.Y.; Vollmuth, P.; Chung, C.; Mansouri, A. AI and Innovation in Clinical Trials. Npj Digit. Med. 2025, 8, 683, doi:10.1038/s41746-025-02048-5.
- Mortlock, T.; Muthirayan, D.; Yu, S.-Y.; Khargonekar, P.P.; Al Faruque, M.A. Graph Learning for Cognitive Digital Twins in Manufacturing Systems. IEEE Trans. Emerg. Top. Comput. 2021, 10, 34–45.
- Glover, D.G.; Hermans, J. Improving the Traceability of the Clinical Trial Supply Chain | Applied Clinical Trials Online Available online: https://www.appliedclinicaltrialsonline.com/view/improving-traceability-clinical-trial-supply-chain (accessed on 22 March 2026).
- Vatankhah Barenji, R.; Hariry, R.E. Blockchain-Enabled Quality by Design System for Clinical Trials. Front. Med. 2025, 12, 1546897, doi:10.3389/fmed.2025.1546897.
- Blockchain in Clinical Trials Market Growth Analysis 2025 to 2034 Available online: https://www.towardshealthcare.com/insights/blockchain-in-clinical-trials-market-sizing (accessed on 22 March 2026).
- Pallmann, P.; Bedding, A.W.; Choodari-Oskooei, B.; Dimairo, M.; Flight, L.; Hampson, L.V.; Holmes, J.; Mander, A.P.; Odondi, L.; Sydes, M.R.; et al. Adaptive Designs in Clinical Trials: Why Use Them, and How to Run and Report Them. BMC Med. 2018, 16, 29, doi:10.1186/s12916-018-1017-7.
- Pezoulas, V.C.; Zaridis, D.I.; Mylona, E.; Androutsos, C.; Apostolidis, K.; Tachos, N.S.; Fotiadis, D.I. Synthetic Data Generation Methods in Healthcare: A Review on Open-Source Tools and Methods. Comput. Struct. Biotechnol. J. 2024, 23, 2892–2910, doi:10.1016/j.csbj.2024.07.005.
- Viceconti, M.; Pappalardo, F.; Rodriguez, B.; Horner, M.; Bischoff, J.; Musuamba Tshinanu, F. In Silico Trials: Verification, Validation and Uncertainty Quantification of Predictive Models Used in the Regulatory Evaluation of Biomedical Products. Methods 2021, 185, 120–127, doi:10.1016/j.ymeth.2020.01.011.
- Chen, W.-A. Drug Supply Chain Optimization for Adaptive Clinical Trials. PhD Thesis, Purdue University, 2019.
- Yue, L.; Xing, S.; Chen, J.; Fu, T. ClinicalAgent: Clinical Trial Multi-Agent System with Large Language Model-Based Reasoning. In Proceedings of the Proceedings of the 15th ACM International Conference on Bioinformatics, Computational Biology and Health Informatics; ACM: Shenzhen China, November 22 2024; pp. 1–10.
- Shi, H.; Zhang, J.; Zhang, K. Enhancing Clinical Trial Patient Matching through Knowledge Augmentation and Reasoning with Multi-Agent 2025.
- Rosenthal, J.T.; Beecy, A.; Sabuncu, M.R. Rethinking Clinical Trials for Medical AI with Dynamic Deployments of Adaptive Systems. Npj Digit. Med. 2025, 8, 252.
- Aguero, D.; Nelson, S.D. The Potential Application of Large Language Models in Pharmaceutical Supply Chain Management. J. Pediatr. Pharmacol. Ther. JPPT Off. J. PPAG 2024, 29, 200–205, doi:10.5863/1551-6776-29.2.200.
- Cunningham, J.W.; Singh, P.; Reeder, C.; Claggett, B.; Marti-Castellote, P.M.; Lau, E.S.; Khurshid, S.; Batra, P.; Lubitz, S.A.; Maddah, M.; et al. Natural Language Processing for Adjudication of Heart Failure in a Multicenter Clinical Trial: A Secondary Analysis of a Randomized Clinical Trial. JAMA Cardiol. 2024, 9, 174–181, doi:10.1001/jamacardio.2023.4859.
- Gargari, O.K.; Habibi, G. Enhancing Medical AI with Retrieval-Augmented Generation: A Mini Narrative Review. Digit. Health 2025, 11, 20552076251337177, doi:10.1177/20552076251337177.
- Liu, S.; McCoy, A.B.; Wright, A. Improving Large Language Model Applications in Biomedicine with Retrieval-Augmented Generation: A Systematic Review, Meta-Analysis, and Clinical Development Guidelines. J. Am. Med. Inform. Assoc. JAMIA 2025, 32, 605–615, doi:10.1093/jamia/ocaf008.
- Liddicoat, J.E.; Lenarczyk, G.; Aboy, M.; Minssen, T.; Porsdam Mann, S. A Policy Framework for Leveraging Generative AI to Address Enduring Challenges in Clinical Trials. NPJ Digit. Med. 2025, 8, 33, doi:10.1038/s41746-025-01440-5.
- 2025 Supply Chain Challenges for the Life Sciences Industry Available online: https://medmarc.com/life-sciences-news-and-resources/publications/2025-supply-chain-challenges-for-the-life-sciences-industry (accessed on 22 March 2026).
- Harmon, D.M.; Noseworthy, P.A.; Yao, X. The Digitization and Decentralization of Clinical Trials. Mayo Clin. Proc. 2023, 98, 1568–1578, doi:10.1016/j.mayocp.2022.10.001.
- DiMasi, J.A.; Smith, Z.; Oakley-Girvan, I.; Mackinnon, A.; Costello, M.; Tenaerts, P.; Getz, K.A. Assessing the Financial Value of Decentralized Clinical Trials. Ther. Innov. Regul. Sci. 2023, 57, 209–219, doi:10.1007/s43441-022-00454-5.
- Goldberg, J.M.; Amin, N.P.; Zachariah, K.A.; Bhatt, A.B. The Introduction of AI Into Decentralized Clinical Trials. JACC Adv. 2024, 3, 101094, doi:10.1016/j.jacadv.2024.101094.
- Oikonomou, E.K.; Thangaraj, P.M.; Bhatt, D.L.; Ross, J.S.; Young, L.H.; Krumholz, H.M.; Suchard, M.A.; Khera, R. An Explainable Machine Learning-Based Phenomapping Strategy for Adaptive Predictive Enrichment in Randomized Clinical Trials. NPJ Digit. Med. 2023, 6, 217, doi:10.1038/s41746-023-00963-z.
- Mikołajewska, E.; Masiak, J.; Mikołajewski, D. Applications of Artificial Intelligence-Based Patient Digital Twins in Decision Support in Rehabilitation and Physical Therapy. Electronics 2024, 13, doi:10.3390/electronics13244994.
- Haripriya, R.; Khare, N.; Pandey, M. Privacy-Preserving Federated Learning for Collaborative Medical Data Mining in Multi-Institutional Settings. Sci. Rep. 2025, 15, 12482.
- Lomurno, E.; Matteucci, M. Federated Knowledge Recycling: Privacy-Preserving Synthetic Data Sharing. Pattern Recognit. Lett. 2025, 191, 124–130.
- Global AI In Clinical Trials: Market Trends & Current Partnerships Available online: https://www.clinicalleader.com/doc/global-ai-in-clinical-trials-market-trends-current-partnerships-0001 (accessed on 22 March 2026).
- Bio Pharma Supply Chain Strategy: Strategic Imperatives & Best Practices Available online: https://feeds.vuroyal.com/blog/bio-pharma-supply-chain-strategy (accessed on 22 March 2026).
About The Author:
 Dr. Hiep X. Nguyen, DPharm, Ph.D., DTM, is Founder and CEO of Novoremedy and a faculty member in Pharmaceutical & Biomedical Sciences at California Northstate University. With over a decade developing topical, transdermal, and injectable products, he holds a Ph.D. from Mercer University and a pharmacy degree from Hanoi University of Pharmacy. A Distinguished Toastmaster and certified pharmacist, he serves as Co-Director of the Vietnamese Association of Pharmacists & Pharmaceutical Scientists, USA, and advises PharmD and PhD students. Dr. Nguyen has authored 30+ peer-reviewed articles, a seminal book (Microneedles: The Future of Drug Delivery), and five book chapters. Recognized among Stanford University’s top 2% of scientists globally, he has delivered numerous international presentations and over 34 scientific abstracts.
Dr. Hiep X. Nguyen, DPharm, Ph.D., DTM, is Founder and CEO of Novoremedy and a faculty member in Pharmaceutical & Biomedical Sciences at California Northstate University. With over a decade developing topical, transdermal, and injectable products, he holds a Ph.D. from Mercer University and a pharmacy degree from Hanoi University of Pharmacy. A Distinguished Toastmaster and certified pharmacist, he serves as Co-Director of the Vietnamese Association of Pharmacists & Pharmaceutical Scientists, USA, and advises PharmD and PhD students. Dr. Nguyen has authored 30+ peer-reviewed articles, a seminal book (Microneedles: The Future of Drug Delivery), and five book chapters. Recognized among Stanford University’s top 2% of scientists globally, he has delivered numerous international presentations and over 34 scientific abstracts.
