CLINICAL SUPPLY PACKAGING ARTICLES
 Inside GCSG 2026: Clinical Supply Impact On Patient Experience
Inside GCSG 2026: Clinical Supply Impact On Patient Experience
This GCSG 2026 event recap with Steve Jacobs (including video) highlights how clinical testing and supply decisions shape patient experience, among other buzzy topics.
CLINICAL SUPPLY PACKAGING RESOURCES
-
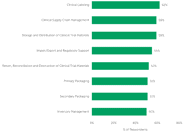
Learn why clinical labeling, clinical supply chain management, and storage and distribution of clinical trial materials received the most votes in a survey of 100 sponsors outsourcing clinical logistics.
-
Learn about Annex VI, which covers changes in the labeling requirements for investigational medicinal products (IMPs) and harmonizes them throughout EU countries.
-
Clinical trials can be completely derailed by the absence of a single component, a lack of clarity in labelling, or errors in temperature-controlled logistics.
-
Discover how the simplification and digitization of clinical supply chain operations enhanced transparency, lowered risks and expenses, and optimized efficiency.
-
As innovative, complex therapeutics advance toward clinical trials, properly handling, storing, and packaging temperature-sensitive critical materials is essential to maintain product integrity.
-
The Daikyo Crystal Zenith nested vials meet the exterior dimensions of the glass standard ISO 8362-1. The vials maintain CCI for up to 6 months and can withstand ultra-cold temperatures and drop conditions.
-
Thinking of packaging and delivery factors early and often will minimize risks to the safety and efficacy of your cell and gene therapy drug product from the bench to the patient.