Expanding Clinical Trials In Africa Without Disruption
By Révérien Uwacu, founder & CEO, Rêve Solutions Consulting

Africa holds 19% of the world's population and carries more than 25% of the global disease burden.1 Yet in 2023, only 1.1% to 4% of clinical trials worldwide were conducted on the continent,2 a figure the World Health Organization describes as a profound structural imbalance. The patient pools are there. The disease profiles are indication rich. The strategic case has never been stronger.
And yet, most sponsors hesitate. Program teams discuss it. Feasibility teams are engaged. And then, quietly, the conversation stalls.
This is not a story about Africa being difficult. It is a story about how the industry approaches the decision to expand and why that approach is broken.
Africa is not an execution problem waiting to be solved. It is a decision problem waiting to be structured.
The fundamental error most sponsors make is skipping directly from interest to execution: commissioning CRO feasibility assessments, requesting logistics quotations, and beginning site selection conversations. All before asking the foundational questions that determine whether the expansion is actually viable, governable, and worth doing. The result is predictable: operational surprises, governance gaps, and programs that retreat from African sites after the first serious deviation.
What follows is a framework-based approach to expanding clinical trials in Africa without disruption, built not on optimism but on structured decision logic applied before a single shipment is planned.

Figure 1 — The Africa clinical trial paradox: disproportionate disease burden, minimal research representation
The Three Uncertainties That Stall African Expansion
Through work with global sponsors across complex emerging market programs, three recurring uncertainties consistently prevent African expansion decisions from moving forward:
- Clinical trial supply (CTS) reliability: cold chain integrity across long and variable corridors, unpredictable customs timelines, and uneven depot and site pharmacy infrastructure.
- Operational governance under deviation: unclear decision authority when excursions occur, slow escalation chains, and the absence of predefined mean time to disposition (MTTD) targets.
- Strategic ROI uncertainty: difficulty quantifying the actual recruitment benefit, unclear regulatory value, and a perception of disproportionate operational risk.
None of these uncertainties is insurmountable. But none of them can be resolved during execution. They must be addressed before the trial begins, ideally before site selection, and certainly before operational commitments are made. This is the gap the industry has not yet filled.
The Missing Layer: Decision Logic Before Execution
The typical sponsor expansion sequence looks like this: a CRO conducts feasibility scoping, a logistics provider is asked to quote, and site selection conversations begin. These are all execution-layer activities. They assume the decision to expand has already been made responsibly. In most cases, it has not.
What is missing is a structured decision layer that sits above execution. One that assesses whether the program is suitable for African corridors, whether those corridors are operationally viable, whether governance structures are in place to handle inevitable deviations, and whether the strategic value justifies the operational complexity.
Without this layer, sponsors enter Africa with optimism rather than evidence. They encounter a deviation – a customs hold in Nairobi, a cold chain excursion at a depot in Lagos, or a replacement batch lead time they did not anticipate and escalate to a sponsor team with no predefined authority matrix, no MTTD target, and no documented risk acceptance. The program stalls. The sites are withdrawn. Africa is labeled as "not ready."
The problem is rarely Africa. The problem is the absence of decision architecture before execution begins.
A Five-Layer Framework For Structured Expansion Decisions
The following framework addresses this gap through five sequential decision layers. Each must be fully assessed before a responsible go/no-go decision can be made. No layer can be bypassed.
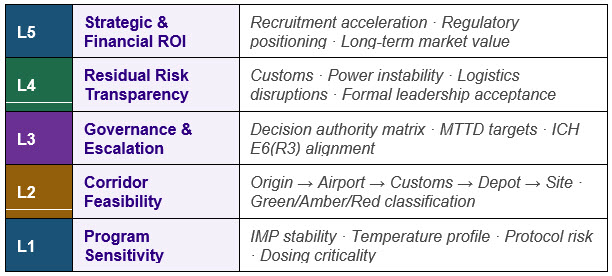
Figure 2 — Five-Layer CTS Africa Expansion Decision Framework: structured before execution begins
Layer 1 — Program Sensitivity Assessment
Before evaluating any African corridor, sponsors must assess whether the program itself can tolerate the operational variability inherent in emerging market supply chains. This is not a judgment on Africa; it is a scientific fitness check that applies to any complex geography.
Key considerations include the IMP's temperature profile and stability window, batch replacement lead times, the protocol's sensitivity to dosing delays, and the clinical consequence of a supply interruption. A highly sensitive biologic with a narrow dosing window and a three-month replacement lead time requires fundamentally different infrastructure than a stable oral tablet with batch flexibility. This layer is often skipped because it feels like a logistics question. It is not. It is a strategic question that determines the scope and cost of everything that follows.
Layer 2 — Corridor Feasibility And Supply Chain Design
Africa is not a single market. It is 54 countries with different customs regimes, infrastructure profiles, cold chain ecosystems, and logistics maturity levels. The question is never whether Africa works, it is whether your specific corridor works.
Each corridor must be assessed end-to-end: origin manufacturing site, export airport, international transport, import customs, regional depot, and clinical site. The assessment covers customs clearance predictability (import permit lead times typically range from four to 16 weeks depending on market3), cold chain handling at airports, validated depot storage with backup power, last-mile logistics with genuine regulatory expertise, and site pharmacy infrastructure.

Figure 3 — Corridor classification framework: Green/Amber/Red assessed end-to-end
Layer 3 — Governance And Escalation Structure
Disruptions will occur. The defining question is not whether a deviation will happen, it is how quickly and clearly the sponsor can respond when it does. This layer defines the governance architecture that enables fast, documented decisions.
The structure includes a predefined decision authority matrix (who has the authority to release IMP after a temperature excursion?), MTTD targets established before site activation, escalation pathways across the site, depot, logistics provider, sponsor QA, and medical monitor and documented deviation management protocols.
Under ICH E6(R3), sponsors bear explicit accountability for the quality of CTS systems, including those operated by CROs and third-party logistics providers.4 A governance framework defined before execution begins is not optional. It is a regulatory expectation and a sponsor protection mechanism.
Layer 4 — Residual Risk Transparency
Even the best-designed supply chains in Africa carry residual risks that cannot be fully eliminated: customs delays due to documentary issues, backup power failures at depots during regional grid instability, transport disruptions, and replacement batch lead times that exceed expectations under manufacturing pressure.
These risks must be formally documented and accepted at the appropriate leadership level before operational commitments are made. This step is consistently skipped, and its absence is one of the primary drivers of program-level governance failures when things go wrong. Formal risk acceptance is not pessimism. It is the sponsor's protection mechanism and the foundation for defensible decision-making throughout the trial.
Layer 5 — Strategic And Financial ROI
Africa expansion must generate tangible, quantifiable value for the development program, not just diversity optics. Three drivers support a strong ROI case.
- Faster patient recruitment. Trial competition in major African markets is significantly lower than in the EU and U.S. Treatment-naive populations and high disease prevalence can materially accelerate enrollment. According to Tufts CSDD research, a single day of clinical development delay represents approximately $800,000 in unrealized prescription drug sales,5 making even modest recruitment improvements significant in value terms.
- Regulatory positioning. The FDA's Food and Drug Omnibus Reform Act (FDORA, 2022) mandated Diversity Action Plans for Phase 3 and pivotal trials, and EMA guidance has consistently emphasized representative populations in regulatory submissions.6 While the regulatory landscape is evolving, the scientific and regulatory rationale for diverse populations remains robust: broader cohorts strengthen approval dossiers, reduce post-marketing commitments, and improve global labeling credibility.
- Long-term market development. Africa's population of approximately 1.4 billion7 represents one of the world's fastest-growing pharmaceutical markets. National health insurance expansion, global health funding programs, and tiered pricing models collectively create a sustainable commercial pathway for sponsors that establish credibility and operational infrastructure early.
What Structured Decision-Making Produces
When all five layers are assessed, sponsors receive a CTS expansion decision pack, sponsor-level evidence base for a defensible go/no-go decision. The pack includes: a program sensitivity assessment; a corridor viability classification by country and route; a governance structure with documented MTTD targets; a formal residual risk register; and a strategic ROI analysis quantifying recruitment, regulatory, and market value.

Figure 4 — Decision output categories from the five-layer assessment
A no-go recommendation does not mean Africa is impossible. It means the current program or corridor configuration does not yet meet the threshold for responsible expansion and it identifies what would need to change in a six to 12-month reassessment window.
Africa expansion becomes a strategic decision — not a speculative discussion.
Practical Implications For Clinical Supply Teams
For clinical supply professionals, the practical implication is clear: advocate for the decision layer before the execution layer begins. When a program team expresses interest in African sites, the first question should not be "Which 3PL do we use?" It should be: "Has this program been assessed for African corridor compatibility?"
Governance structures need to be built before site activation, not assembled during a deviation. MTTD targets need to be set in advance, not negotiated under pressure. Residual risks need to be on a leadership-level register before the first IMP shipment lands at an African airport.
The 2024 Access to Medicine Index found that only 27.5% of late-stage R&D projects from 20 top pharmaceutical companies included one or more African country.2 This underrepresentation is not primarily a scientific problem. It is a decision infrastructure problem and clinical supply professionals are uniquely positioned to solve it.
Conclusion
Africa is ready for more clinical trials. The patient populations are there. The disease profiles align with global development pipelines. The regulatory environment is maturing. And the strategic case – recruitment speed, regulatory diversity, long-term market positioning – is quantifiable and compelling.
What has been missing is not execution capability. It is decision architecture. Sponsors that build a structured decision framework before execution begins will enter Africa with realistic expectations, documented risk acceptance, predefined governance, and the confidence that comes from evidence rather than optimism.
They will also be the ones still running those trials when the first deviation arrives because they will already know exactly what to do.
References:
- World Health Organization. A Heavy Burden: The Productivity Cost of Illness in Africa. WHO Regional Office for Africa, 2019. Available at: https://www.afro.who.int/publications/heavy-burden-productivity-cost-illness-africa
- Access to Medicine Foundation. Harnessing Africa's Untapped Clinical Trial Potential. September 2025. Available at: https://accesstomedicinefoundation.org/insights-resources/access-insights/harnessing-africas-untapped-clinical-trial-potential [citing WHO clinical trial registration data, 2023]
- Uwacu, R. CTS Africa Expansion Decision Framework. Rêve Solutions Consulting, 2025. Import permit lead time ranges derived from corridor assessments conducted across multiple African markets including West, East, and Southern Africa corridors.
- International Council for Harmonisation. ICH E6(R3) Guideline for Good Clinical Practice. Step 4, May 2025. Available at: https://www.ich.org/page/efficacy-guidelines
- Smith Z, DiMasi J, Getz K. New Estimates on the Cost of a Delay Day in Drug Development. Therapeutic Innovation & Regulatory Science. 2024. DOI: 10.1007/s43441-024-00667-w
- U.S. Food and Drug Administration. Diversity Action Plans to Improve Enrolment of Participants from Underrepresented Populations in Clinical Studies: Draft Guidance for Industry. June 2024. Available at: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/diversity-action-plans-improve-enrollment-participants-underrepresented-populations-clinical-studies
- United Nations, Department of Economic and Social Affairs. World Population Prospects 2024. Available at: https://population.un.org/wpp/
About The Author:
 Révérien Uwacu is the founder & CEO of Rêve Solutions Consulting, where he advises pharmaceutical and biotech companies on clinical trial supply chain strategy and global expansion into emerging markets, with a focus on Africa. He is the creator of the CCIM (Country-Contextualised IMP Management) framework, a strategic model designed to de-risk and accelerate clinical trial execution in underrepresented regions.
Révérien Uwacu is the founder & CEO of Rêve Solutions Consulting, where he advises pharmaceutical and biotech companies on clinical trial supply chain strategy and global expansion into emerging markets, with a focus on Africa. He is the creator of the CCIM (Country-Contextualised IMP Management) framework, a strategic model designed to de-risk and accelerate clinical trial execution in underrepresented regions.
Révérien will be speaking at the Clinical Trial Supply Forum (May 2026) and the GCSG European Knowledge Forum 2026 (October 2026), contributing to industry discussions on supply chain innovation, patient diversity, and market expansion. He can be reached at info@reve-solutions.com.
