The Hidden Supply Chain Risks That Can Derail Small Biotech Trials
By Bob Lozito, MBA, MA

Small biotechnology companies are at the forefront of developing groundbreaking therapies in oncology, rare diseases, and advanced modalities like cell and gene therapies and antibody-drug conjugates (ADCs). However, their clinical trial supply chains — from raw material procurement through patient dosing — represent a critical vulnerability that can derail programs and exhaust limited capital. Operating with typically $10 million to $50 million in funding post-Series A, teams of fewer than 20 people, and monthly burn rates of $2 million to $5 million, these firms face industry-leading rates of drug waste (50%-75%), trial delays averaging six to 12 months, and runway depletion 20%-30% faster due to supply chain failures. This comprehensive paper examines both the core challenges (demand forecasting, vendor silos, cold chain logistics, inventory waste, and quality gaps) and second-order issues (upstream raw materials, tech transfers, data fragmentation, trial design complexity, and organizational constraints), analyzing root causes, impacts, and practical mitigation strategies tailored to resource-constrained environments.1,2,5,7,8,10,11,12
Introduction
The clinical trial supply chain in biotechnology encompasses the end-to-end process of manufacturing, packaging, labeling, distributing, and managing investigational medicinal products (IMPs) to ensure patients receive the right dose at the right time. For small biotechs, this chain is disproportionately complex due to novel modalities requiring specialized handling, global multisite trials, and razor-thin margins for error. Unlike Big Pharma with established infrastructure, small firms outsource 80%-90% of execution to contract development and manufacturing organizations (CDMOs), contract research organizations (CROs), and logistics providers, creating coordination nightmares. A single disruption, such as an inaccurate forecast or a cold chain failure, may consume between 20% and 40% of a program's budget and threaten critical venture capital milestones including IND clearance or proof-of-concept data.2,3,4,6,7,8,9
Core Supply Chain Challenges
Demand Forecasting Uncertainty
The cornerstone of clinical supply management is accurate demand forecasting, which depends on predicting patient enrollment curves, site activation timelines, dropout rates (typically 15%-25%), and protocol amendments that affect nearly one-third of trials. Small biotechs frequently rely on rudimentary spreadsheet models derived from historical averages, such as one-half to one patient per site per month, without accounting for trial-specific factors like adaptive designs, regional accrual differences (slower in Europe versus the U.S.), or external disruptions like competing studies or pandemics. For biologics with manufacturing lead times of 12 to 18 months and shelf lives ranging from six to 24 months, forecasting errors can have significant consequences: over-forecasting may result in substantial losses due to expired inventory, while under-forecasting can cause site stockouts, interrupt dosing schedules, and diminish investigator confidence.1,4,7,8,9,14
Lean teams lacking dedicated supply chain expertise exacerbate the problem. In a typical Phase 2 oncology trial planning for 200 patients, actual enrollment might reach only 120 due to recruitment challenges in Eastern Europe, leading to $5 million to $10 million in wasted GMP material. The impact extends beyond costs: stockouts increase patient dropouts and CRO demotivation, directly threatening value-inflection milestones that unlock subsequent VC tranches. Mitigation requires early deployment of randomization and trial supply management (RTSM) systems for real-time visibility and automated resupply triggers, coupled with Monte Carlo simulations incorporating probability-of-success benchmarks. 4,7,8,14
Siloed Decision-Making And Vendor Misalignment
Clinical supply chains typically involve five to 15 external partners, including CROs for trial execution, CDMOs for manufacturing, and logistics firms for distribution. Without integration, CMC teams produce batches oblivious to real-time clinical enrollment data, while protocol amendments necessitate expensive relabeling and repackaging. Small biotechs, with their one- or two-person "departments," lack supplier relationship management (SRM) or project management office (PMO) functions, resulting in fragmented data across Excel files, vendor portals, and email threads. A common failure mode is a CDMO delivering non-QP certified material, delaying qualified person for batch release by weeks and stalling site activations.2,6,8,9
These silos cause trial delays of three to six months and cost overruns of 15%–25%, with regulatory audits highlighting gaps in accountability. Effective countermeasures include selecting integrated CRO-CDMO hybrids for shared responsibility, negotiating master service agreements (MSAs) with strict service level agreements (SLAs) for handoffs, and implementing unified platforms that consolidate data flows.2,3,4,9
Cold Chain And Logistics Vulnerabilities
Approximately 70% of biotech assets — monoclonal antibodies, ADCs, and cell/gene therapies — demand ultra-low temperature storage (minus 80 degrees C to minus20 degrees C), making the supply chain exquisitely sensitive to excursions, customs delays, and dry ice shortages. Global trials compound risks with varying import regulations, language-specific labeling, and the rise of direct-to-patient (DTP) models that introduce chain-of-custody challenges. Budget-constrained small biotechs often forgo redundancies like backup depots or validated specialist shippers, leaving them exposed: a single minus5 degree C deviation can destroy a $2 million batch.1,2,9
Enrollment slowdowns of up to 30% result, particularly in regions like Asia-Pacific where customs holds are routine. Proactive strategies involve failure modes and effects analysis (FMEA) mapping pre-trial, GPS-enabled refrigerated shippers, establishment of three to five regional depots, and 20% contingency buffers validated through thermal mapping.1,9,13
Inventory Imbalance And Waste
Biotech manufacturing yields vary widely (70%-90% is typical), and conservative buffering against uncertainty drives industry-standard waste rates of 55%-75%. Short shelf lives necessitate just-in-time production, but protocol amendments render kits obsolete, while lack of dual-sourcing heightens single-vendor dependencies. For small firms, this translates to $10 million to $20 million lost per program, shortening runways from 24 to 18 months and forcing premature bridge financings.1,7,8
Optimization via dynamic RTSM algorithms targeting 95% service levels, combined with AI-driven point-of-sale forecasting tools that boost accuracy by 44%, offers substantial relief. Unblinded surplus can often be donated or repurposed for toxicology studies.4,14
Regulatory And Quality Management Gaps
Managing GMP, GDP, and GCP compliance for international vendors can be too much for part-time QA staff. Inspections frequently flag incomplete stability data, unaudited subcontractors, or data integrity issues, triggering clinical holds. Small biotechs delay full quality management system (QMS) implementation until crises arise, amplifying remediation costs to 10%-20% of budgets and adding six+ months to timelines. Consultants for vendor audits and embedding quality requirements in MSAs provide a scalable path forward.2,6,9
Second-Order And Emerging Challenges
Beyond these foundational issues, small biotechs encounter upstream data-related, design-driven, and structural hurdles that silently amplify risks. Upstream vulnerabilities arise from raw material shortages of key inputs such as chromatography resins, single-use systems, cell culture media, and specialized packaging. These shortages are compounded by long lead times of six to 12 months and reliance on single suppliers, largely because purchasing power is limited. Tech transfer failures — stemming from incomplete bills of materials (BOMs) or equipment mismatches at receiving CDMOs — routinely delay scale-up by two to four months. Numerous organizations certify vendors just for Phase 1, but later encounter issues when it comes to Phase 3 capacity or serialization requirements, which then leads to expensive transitions and the need to revalidate.5,10,11
Data fragmentation across unstructured sources hampers analytics, QP releases, and predictive planning, while ad hoc risk management without formal FMEA or Supply Chain Operations Reference (SCOR) models leaves firms reactive. Lean teams suffer expertise gaps in GxP nuances, often missing trial design flaws like overly complex visit schedules that strain supply. Trial complexities such as adaptive protocols, frequent amendments, decentralized models, comparator sourcing, and co-medications introduce labeling, blinding, and custody intricacies. Novel modalities like ADCs and cell/gene therapies multiply manufacturing sites and process windows, elevating failure rates. Organizationally, investor pressure fosters milestone myopia — prioritizing speed over robustness — and treats supply as tactical rather than strategic, lacking dedicated ownership. Global escalation in requirements like GS1 serialization and data integrity further strains capacities.2,5,7,9,10,11,12,16,17
Comparative Impact Analysis
The table below illustrates how challenges evolve by phase, highlighting small biotech vulnerabilities and budget risk.1,4,9,13
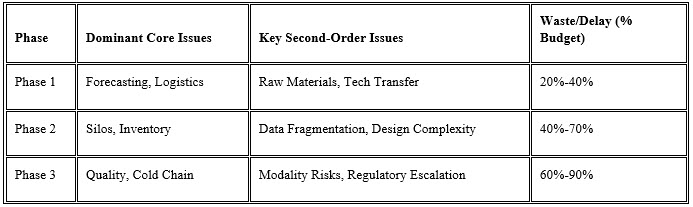
Strategic Recommendations And Conclusion
Small biotechs should embed supply strategy into trial design via pre-IND cross-functional charters defining KPIs like <5% stockouts and <20% waste. Allocate 5%-10% of budgets to transformative technologies — RTSM integrated with AI analytics — for triple ROI through waste reduction. Cultivate ecosystems of two or three trusted vendors via MSAs with performance escalators, hire or contract fractional supply chain leaders, and invest in training through organizations like ISPE (International Society for Pharmaceutical Engineering). Proactive monitoring with weekly dashboards and annual FMEA refreshes ensures agility.3,4,6,15
Ultimately, mastering clinical supply chains transforms a perennial liability into a competitive differentiator. Firms achieving integration extend runways by 25%, reliably hit milestones, and command Series B valuations at 20%-30% premiums, positioning them for sustainable growth toward midsize operations.2,6,8,12
Key Takeaways
Small biotech firms face severe clinical trial supply chain risks that can waste 50%-75% of materials, delay trials by six to 12 months and deplete funding 20%-30% faster. Core issues include inaccurate demand forecasting from spreadsheets ignoring dropouts and amendments, siloed vendors lacking integration, cold chain failures destroying multimillion-dollar batches, high inventory waste, and quality gaps. Second-order challenges involve raw material shortages, tech transfer errors, data silos, complex trial designs, and lean teams without supply expertise.
Mitigation Strategies
- Deploy RTSM systems early with AI forecasting and Monte Carlo simulations for 95% service levels and 44% accuracy gains.
- Use integrated CRO-CDMO partners, MSAs and SLAs, and unified data platforms to cut delays by three to six months.
- Create redundancies with regional depots, GPS tracking, and FMEA for cold chain; dedicate 5%–10% of the budget to tech for triple ROI.
- Embed supply planning in trial design with KPI (< 5% stockout, < 20% waste) and fractional supply chain leaders.
Mastering these turns liabilities into advantages, extending runways 25% and boosting valuation 20%-30%.
References:
- Medmarc. (2025, October 6). 2025 Supply Chain Challenges for the Life Sciences Industry. Medmarc.com. https://medmarc.com/life-sciences-news-and-resources/publications/2025-supply-chain-challenges-for-the-life-sciences-industry
- Pronav Clinical. (2025, March 11). Top 5 Challenges in Clinical Trial Supply Chain Operations (and ...). Pronavclinical.com. https://www.pronavclinical.com/post/top-5-challenges-in-clinical-trial-supply-chain-operations
- N-Side. (2025, August 10). Tips and tricks for an efficient clinical supply chain. N-side.com. https://www.n-side.com/en/insights/no-slider-tips-and-tricks-for-an-efficient-clinical-supply-chain/
- TrialZen. (2024, June 10). How Forecasting and Optimization Work in Clinical Trial Supply. Trialzen.com. https://www.trialzen.com/blog/how-forecasting-and-optimization-work-in-clinical-trial-supply-management
- DesPharma Consulting. (2025, August 31). Navigating Pharmaceutical Supply Chain Challenges: 2026 Risk .... Despharmaconsulting.com. https://despharmaconsulting.com/navigating-pharmaceutical-supply-chain-challenges-2026-risk-mitigation-strategies-for-biotech-in.
- Siron Clinical. (2025, March 4). How Small Biotech Can Overcome Clinical Trial Hurdles. Sironclinical.com. https://sironclinical.com/how-small-biotech-can-overcome-clinical-trial-hurdles/
- LinkedIn Pulse. (2024, June 2). Only 15% of Clinical Trials Overcome These Supply Chain Challenges. Linkedin.com. https://www.linkedin.com/pulse/only-15-clinical-trials-overcome-supply-chain-challenges-4xewe
- N-Side. (2022, June 6). The 3 biggest challenges in the clinical trial supply chain. N-side.com. https://www.n-side.com/en/insights/the-3-biggest-challenges-in-the-clinical-trial-supply-chain/
- Experic Services. (2025, May 8). Managing the Key Challenges of Clinical Trial Supply Services in .... Expericservices.com. https://expericservices.com/managing-the-key-challenges-of-clinical-trial-supply-services-in-complex-studies/
- BioProcess International. (2024, July 30). Supply-Chain Challenges in Biotechnology. Bioprocessintl.com. https://www.bioprocessintl.com/supply-chain/supply-chain-challenges-in-biotechnology
- CAS. (2025, September 4). Creating resilience and efficiency in biotech supply chains. Cas.org. https://www.cas.org/resources/cas-insights/biotech-supply-chain
- Pharmaceutical Executive. (2025, October 6). Helping Small Biotechs & Startups Navigate Complexities and .... Pharmexec.com. https://www.pharmexec.com/view/helping-small-biotechs-startups-navigate-complexities-accelerate-time-market
- Clinical Supply Leader. (2026, January 22). Managing Supply Chain Risk In Scale-Up And Multisite Trials. Clinicalsupplyleader.com. https://www.clinicalsupplyleader.com/doc/managing-supply-chain-risk-in-scale-up-and-multisite-trials-0001
- Ozmosi. (2025, October 15). How Next-Gen POS Forecasting Can Improve Clinical Trial Ozmosi.com. https://www.ozmosi.com/next-gen-pos-forecasting-can-improve-clinical-trial-accuracy-by-44/
- Journal for Clinical Studies. (2024). Forecasting and Kit Design in Clinical Trials for the Pharmaceutical ... [PDF]. https://journalforclinicalstudies.com/wp-content/uploads/2024/10/Forecasting-and-Kit-Design.pdf
- Thermofisher. (2024, November 4) Survey: Highlighting Drug Development Challenges & Opportunities https://www.ppd.com/blog/drug-development-challenges-opportunities-2024/
- Fiercebiotech. (2025 May, 29) How Merck Research Labs is Evolving its Global Clinical Supply to Meet the Demands of Newer Modalities https://www.fiercebiotech.com/editorial/how-merck-research-labs-is-evolving-its-global-clinical-supply-to-meet-the-demands-of-newer-modalities
About The Author:
 Robert J. (Bob) Lozito is a seasoned pharmaceutical professional with more than 30 years of leadership in global clinical supply chains and clinical trial operations. Known for his strategic clarity, operational rigor, and collaborative style, he has guided major pharma and biotech organizations through complex regulatory, logistical and compliance challenges. Bob became known for his systematic approach to solving problems, his compassionate communication style, and his ability to guide and uplift teams with careful attention and clear direction. Beyond his career, he aims to create a legacy, support charities, and positively influence his community. His actions reflect the principles of perseverance, integrity, and service that have shaped him throughout his life.
Robert J. (Bob) Lozito is a seasoned pharmaceutical professional with more than 30 years of leadership in global clinical supply chains and clinical trial operations. Known for his strategic clarity, operational rigor, and collaborative style, he has guided major pharma and biotech organizations through complex regulatory, logistical and compliance challenges. Bob became known for his systematic approach to solving problems, his compassionate communication style, and his ability to guide and uplift teams with careful attention and clear direction. Beyond his career, he aims to create a legacy, support charities, and positively influence his community. His actions reflect the principles of perseverance, integrity, and service that have shaped him throughout his life.
