The NDC-12 Ripple Effect: Why Clinical Trial Supply Chains Must Act Seven Years Early
By Manish Kumar Garg, associate director, IT Applications, Hikma Pharmaceuticals

The most dangerous phrase in clinical supply is, “that’s a commercial problem.”
I’ve heard it too many times. And every time I do, I know we’re about to watch something complicated get unnecessarily worse.
The NDC-12 transition — the FDA’s mandate to move to a standardized 12-digit National Drug Code (NDC) in a uniform 6-4-2 format, effective March 7, 2033 — is being handled exactly that way in too many organizations right now.1 It gets routed to labeling teams. Assigned to commercial operations. And clinical supply leadership nods along, satisfied that it’s someone else’s agenda item.
That instinct is understandable. But identifier misalignment causes real operational damage — during DSCSA, during COVID-era supply rebuilds, during late-phase tech transfers that hit unexpected walls. This is the moment to push back.
This is not a labeling compliance exercise. It is a product identity decision. And product identity is not created at launch. It is created for the first time a product enters a trial system. (See fig1 for lifecycle reference)
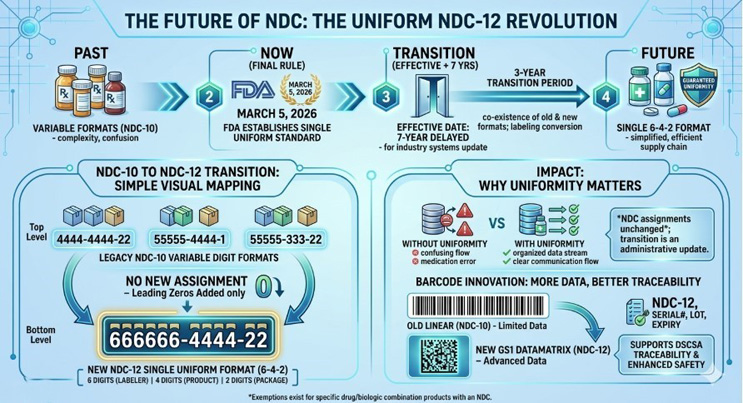
The Operating Environment Has Fundamentally Changed
Before the pandemic, identifier inconsistencies between clinical and commercial systems were the kind of thing you sorted out during tech transfer — painful but bounded. That operating model is gone. The pandemic accelerated an evolution the industry had been slowly resisting. Direct-to-patient shipments scaled dramatically. Decentralized trial models became operationally mainstream, with estimates suggesting 40%–45% of studies incorporated decentralized elements by 2022, up from roughly 20% just a year earlier.2 Real-time inventory visibility stopped being a nice-to-have and became a survival requirement as global logistics fractured. Regulatory expectations around data integrity tightened in ways that didn’t loosen afterward. The FDA formalized its position on decentralized clinical trials in 2023 and 2024, establishing framework expectations for data traceability and remote operations that are now part of study design from the outset.3,4 RTSM platforms, once configured primarily around site-based dispensing, have had to integrate direct-to-patient capabilities, adaptive supply forecasting, and cross-border logistics in ways that create far more complex data dependencies than existed before the pandemic.5,6
In this environment, a product’s identity is no longer just a label or a field in a database. It’s a thread that must run intact through manufacturing, trial operations, partner systems, and regulatory submissions — across geographies, through the full life cycle.
NDC-12 isn’t introducing that requirement. It’s exposing where we haven’t yet met it.

The Transition Window Creates A Structural Collision Most Teams Aren’t Seeing
The FDA’s rule includes a coexistence period where legacy 10-digit and new 12-digit codes will both be valid — running from March 7, 2033 through March 6, 2036.7 That sounds like a commercial timing issue until you map it against a clinical timeline.
A trial initiated today will likely still be running when that window opens. Its IRT system was configured at study start-up with product identifiers that map to the 10-digit structure. Manufacturing scale-up is proceeding with commercial CMOs that are already moving toward NDC-12 in their own systems. When the product approaches approval, the commercial identifier will have shifted, while clinical systems still reference the earlier format.
At that point, reconciliation is no longer a data conversation. It becomes a constraint embedded inside operating systems that were never designed to absorb the change.
We saw a version of this during DSCSA implementation. Organizations that treated serialization as a downstream requirement found themselves requalifying packaging lines and rebuilding data flows during tech transfer.8,9 The lesson was clear: when you don’t align identity early, you pay for it later in the most expensive way possible — when you can least afford the time.

A Four-Phase NDC-12 Readiness Framework
What separates organizations that navigate transitions like this smoothly from those that don’t isn’t technology. It’s sequencing. Here is a practical phased approach matched to how clinical supply programs operate:
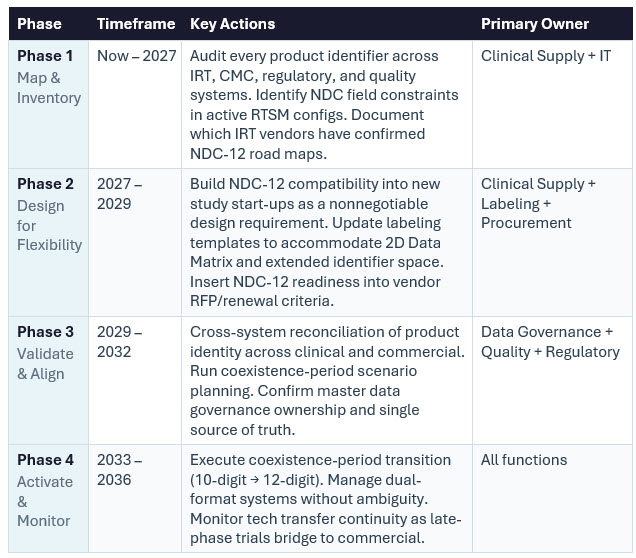
Each phase builds on the one before. Phase 1 failures cascade into Phase 3 crises. The organizations that will have the easiest 2033 are the ones executing Phase 1 now.
Systems Configured Early Are Lived with For Years
IRT and RTSM platforms are configured at study start-up by design — built around patient safety, trial integrity, and regulatory compliance.6,10 You don’t change foundational system structure mid-study.
Which means a field constraint set in 2024 is still a field constraint in 2030. A validation rule written with a specific identifier format in mind will quietly reject a different format when the transition hits. Mapping tables that made sense under the old structure require manual intervention. Integrations built on a specific data architecture expose themselves at exactly the moment when clinical and commercial teams need to be speaking the same language.
The pattern is almost always the same: a small design decision made at system start-up, made under time pressure, without the 2033 transition on anyone’s radar, turns into a structural constraint that takes months and significant resources to navigate around.

Labeling: Where Physical And Digital Identity Actually Intersect
Clinical labeling gets treated as a technical execution function. It is actually one of the highest-leverage decision points in the entire life cycle. Global trials require coordination across languages, regulatory jurisdictions, packaging sites, and quality systems. Once a label design is established and validated, changing it is not a quick exercise — the timelines alone make late-stage redesign genuinely painful. The NDC-12 rule also updates barcode requirements, permitting both linear and nonlinear formats — including 2D Data Matrix barcodes — provided they conform to internationally recognized standards such as those developed by GS1.1 Critically, for products subject to both NDC labeling and DSCSA product identifier requirements, a single 2D data matrix barcode may satisfy both beginning March 7, 2033.7 What looks like a compliance formatting update is, in practice, the moment where physical product packaging and digital data architecture either align or don’t.

Master Data: The Conversation No One Wants To Have
Every organization has some version of this problem. Product identity exists in multiple places — clinical systems, manufacturing systems, regulatory systems, quality systems — and those places don’t always agree with each other. When identifiers are stable, the inconsistency is manageable. When identifiers change, the fragmentation becomes visible in the worst possible circumstances.
McKinsey’s research on pharmaceutical supply chain resilience has identified three foundational ingredients that separate the most resilient organizations from the rest: end-to-end visibility, high-quality master data, and effective scenario planning .11 The persistent gap is not typically in visibility technology — it’s in the underlying data governance. Their 2024 follow-on survey reinforced this: only a quarter of companies have formal, board-level processes for supply chain risk, and even fewer have proactive governance frameworks for data model changes.12
The question NDC-12 forces is one that has been deferred for years in most organizations: what is the single source of truth for product identity, and who is accountable for it? That’s not a technology question. That’s an organizational one.
When governance is unclear, alignment happens reactively — during tech transfer, during an audit, during a crisis. When governance is established, alignment becomes part of design. The organizations using this transition to define that ownership clearly are building infrastructure that will pay off across every product life cycle transition that follows.

This Is A Clinical Design Decision, Not A Compliance Deadline
The organizations that will have the hardest time with NDC-12 aren’t the ones with the weakest technology. They’re the ones that categorized this as a future commercial problem and built clinical systems in the intervening years without it on their radar.
For those organizations, the transition will arrive as a disruption layered on top of complexity. For the ones that acted early, it will arrive as an extension of what they’ve already built. That gap isn’t about resources or ambition. It’s about when you recognize what this actually is.
What looks like a commercial compliance requirement is, at its core, a clinical design decision. The moment to make that decision is now, not when the transition window opens, and certainly not when you’re already inside a study that can’t easily absorb the change.
Seven years feels like a long runway. It isn’t — not when you account for study timelines, system configuration cycles, label validation, and the organizational work of establishing data governance that didn’t exist before. The FDA has signaled clearly that it does not intend to extend the deadline.13
McKinsey research in line with the strategy/insights in the article to emerge from a disruption like NDC12 identifier transformation. 14
The organizations that navigate transitions like this successfully aren’t the ones that moved fastest at the end. They’re the ones that started thinking about it first.
References:
APA 7th Edition
- U.S. Food and Drug Administration. (2026a, March 5). Revising the National Drug Code Format and Drug Label Barcode Requirements [Final rule]. Federal Register, 91(43). https://www.federalregister.gov/documents/2026/03/05/2026-04368/revising-the-national-drug-code-format-and-drug-label-barcode-requirements
- IntuitionLabs. (2025, November). Patient-centric RTSM: Enhancing clinical trial experience through innovation. https://intuitionlabs.ai/articles/patient-centric-rtsm-enhancing-clinical-trial-experience
- U.S. Food and Drug Administration. (2024). Decentralized clinical trials for drugs, biological products, and devices: Guidance for industry. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/decentralized-clinical-trials-drugs-biological-products-and-devices
- Stern, A. D., & Trusheim, M. (2024, February 9). Decentralized clinical trials and digital health technologies. In Transformation of the clinical trial enterprise: Lessons learned from the COVID-19 pandemic (Chapter 4). Office of the Assistant Secretary for Planning and Evaluation (ASPE). https://www.ncbi.nlm.nih.gov/books/NBK609002/
- Rickenbacher, E. (2021, October 4). The state of IRT and RTSM technologies [Interview]. Applied Clinical Trials Online. https://www.appliedclinicaltrialsonline.com/view/the-state-of-irt-and-rtsm-technologies
- Signant Health. (2025, April). RTSM and IRT in clinical trials: Essential guide. https://signanthealth.com/resources/blog/rtsm-irt-clinical-trials-guide
- U.S. Food and Drug Administration. (2026b). National Drug Code Format. https://www.fda.gov/drugs/electronic-drug-registration-and-listing-system-edrls/national-drug-code-format
- Mackey, T. K., & Liang, B. A. (2019). Combating substandard and counterfeit medicines by securing the pharmaceutical supply chain: The Drug Supply Chain Security Act (DSCSA) of 2013. PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC6438546/
- U.S. Food and Drug Administration. (2023). Drug Supply Chain Security Act (DSCSA). https://www.fda.gov/drugs/drug-supply-chain-integrity/drug-supply-chain-security-act-dscsa
- Oracle Life Sciences. (2024). Modernizing clinical trials: The role and significance of RTSM. https://www.oracle.com/life-sciences/modernizing-clinical-trials-role-and-significance-of-rtsm/
- McKinsey & Company. (2023, November). Tech and regionalization bolster supply chains, but complacency looms: 2023 Supply Chain Pulse Survey. https://www.mckinsey.com/capabilities/operations/our-insights/tech-and-regionalization-bolster-supply-chains-but-complacency-looms
- McKinsey & Company. (2024, October). McKinsey Global Supply Chain Leader Survey 2024: Vulnerabilities remain despite progress. https://www.mckinsey.com/capabilities/operations/our-insights/supply-chain-risk-survey-2024
- Faegre Drinker Biddle & Reath LLP. (2026, March). FDA finalizes rule requiring 12-digit National Drug Code (NDC). https://www.faegredrinker.com/en/insights/publications/2026/3/fda-finalizes-rule-requiring-12-digit-national-drug-code-ndc
- McKinsey & Company. (2022, October). Emerging from disruption: The future of pharma operations strategy. https://www.mckinsey.com/capabilities/operations/our-insights/emerging-from-disruption-the-future-of-pharma-operations-strategy
About The Author:
 Manish Kumar Garg is associate director, IT Applications at Hikma Pharmaceuticals, with 20+ years of experience driving digital transformation in life sciences. He leads global serialization, track & trace, and supply chain applications, with deep expertise in DSCSA compliance, data integrity, and enterprise platforms. Manish has a proven track record in regulatory programs, technology modernization, and cross-industry collaboration with SAP Co-innovations, GS1, HDA, and TraceLink. An active industry speaker and thought leader, he focuses on transforming compliance into strategic value through visibility, trust, and intelligent supply chains. He can be contacted at manishgrg@gmail.com.
Manish Kumar Garg is associate director, IT Applications at Hikma Pharmaceuticals, with 20+ years of experience driving digital transformation in life sciences. He leads global serialization, track & trace, and supply chain applications, with deep expertise in DSCSA compliance, data integrity, and enterprise platforms. Manish has a proven track record in regulatory programs, technology modernization, and cross-industry collaboration with SAP Co-innovations, GS1, HDA, and TraceLink. An active industry speaker and thought leader, he focuses on transforming compliance into strategic value through visibility, trust, and intelligent supply chains. He can be contacted at manishgrg@gmail.com.
